Saturday one of the senior Chemistry lecturer Mr David Loh brief us about the upcoming practical test for AS level. It will be my batch's first exam on the 15th May, it's just 2 weeks from the D-day....
Here I'll sum up some of the important aspects Mr Davidson covered during his briefing.
NOTE: There's lots of chemistry terms and symbols below, which might confuse those who aren't taking Chemistry. Proceed at your own "risk", heh.
Preparing for the test
- Candidates are required to bring their lab coat, goggle, exam registration slip, identity card and calculator to the exam. Also, candidates must present at the lab 15 mins prior the actual exam time to avoid any delays.
- Check the instruments and apparatus before starting the exam. Any damage or leakage must be reported to the examiner as soon as possible.
- Remember, practical paper takes up 23% of the AS Chemistry paper. It is the paper to determine whether you'll be getting an A or not.
Titration
- A white tile is provided for better observation to spot the colour change during titration.
- The burette and pipette have to be rinsed with the solution before use to avoid any contamination and inaccuracy measurement of the solutions used.
- Value for rough titration is always greater than accurate titration.
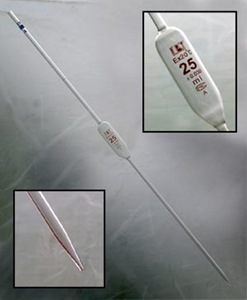
Burette & Pipette
- Do not place the practical report too close to the titration setup to avoid the solutions spilled onto the paper.
- Example table to record titration readings:
| Titration | Rough | Accurate | ||
| 1 | 2 | 3 | ||
| Final /cm3 | ||||
| Initial /cm3 | ||||
| Volume /cm3 |
Note: At least 2 readings with the margin ≤ 0.10 cm3 are taken as the experiment result.
Salt analysis - testing for the ions
- Adding 1cm3 reagent is considered "equal amount". Measurement more than 2cm3 is considered excess.
- Take notes of the cations Pb2+, Al3+ & Zn2+. These 3 amphoteric ions exhibits special observations in order to deduce the ions. The ions will form white ppt upon adding NaOH drop by drop, and will dissolve in excess NaOH to form a clear solution. When the solution is treated with HNO3, the white ppt will form again. This test is only valid for the 3 ions as mentioned above. The reason behind this is because the hydroxide ions form are insoluble white ppt, and the hydroxide will further react with excess OH- to form a complex ions which are soluble. This can be referred to Le Chatelier's principle.
- Pb2+ can be distinguished from Al3+ by reacting with Cl- to form PbCl2, an insoluble solution.
- Some high-charged ions, eg: Al3+, Fe3+ & Cr3+ can exhibit acidic properties, especially when reacting with CO32- to form CO2 gas.
- Transition element ions, eg: Cu2+, Fe2+, Cr3+ & Mn2+ are coloured solutions. Therefore it's easy to deduce the ions based on their significant colours, for example, Cu2+ is a blue solution.
From left to right, aqueous solutions of: Co(NO3)2 (red); K2Cr2O7 (orange); K2CrO4 (yellow); NiCl2 (green); CuSO4 (blue); KMnO4 (purple).
- HCl is a useful reagent to test for certain ions. If there's no gas evolved when HCl is added, one can deduce that SO32-, NO2- and CO32- is absent. These 3 anions will react with HCl and gases will evolve. The gases evolved are SO2, NO2, & CO2 respectively. Test for the gases will be listed below.
Test for gases:
| Gas | Test | Observation |
| CO2 | React with acid (CO32-) | Effervescence/ bubbles evolved. Gas turns limewater cloudy. |
| NH3 | Heating (NH4+) | Pungent gas evolved. Turn damp red litmus paper blue. |
| SO2 | React with acid (SO32-) | Colourless gas with smoking smell, turns K2Cr2O7 from orange to green. (smells like burning matchstick) |
| NO2 | React with acid (NO2-) | Brown gas evolved with pungent smell. |
| Cl2 | Yellowish-green gas. Turns damp blue litmus paper red, and bleaches it. |
Note: When heating a solution, it's best to prepare 2 damp litmus papers (red & blue) and place it on top of the boiling tube when heating to test the gas evolved.
- AgNO3 is the common reagent to test for halides: Cl-, Br-, I-. Observations are white ppt, pale cream ppt and yellow ppt respectively.
- Heating a solution with Al foil is to test NO3- & NO2-.
- Ba2+ is the reagent to test SO42- & SO32-.
- It is possible that the salt analysis part is replaced by organic compounds tests.
Calculations
- During calculations, answers which haven't been finalised may be written in 4 or 5 sf (significant figures). Only the final answer for the question need to be rounded up to 2sf or 3sf.
- If unsure with the significant figures needed for the answer, put 3sf instead.
- Approximation sign (≈) cannot be used in answering the question unless asked to do so.
- Reading from burette is always written in 2 decimal places, eg: 25.75 cm3. Note that the 2nd decimal number must be either 0 or 5.
- Pipette reading is fixed at 25.0 cm3.
- Thermometer reading is usually written in 1 decimal place figure, eg: 35.4°C.
- Types of errors:
- Systematic error: faulty instruments giving zero error (thermometer, stopwatch).
- Random error: error due to the surroundings (wind, heat).
- Estimated error for burette reading = half of the smallest division = 0.05cm3.
- Oxidation states and ΔH (kJ mol-1) must write + or - .
- It is possible to answer the questions based on understandings of the Chemistry outside the syllabus, and you may use any methods to answer the question as long as the Chemistry is correct. In other words, you can answer this Pre-U level questions with your post-grad level understanding if you like.
I think this basically sums up the 2 hours briefing session. I hope it helps not just me but those who're taking the exam too.
[UPDATE]: A presentation I've made for identifying cations. You can view the slide here.


No comments:
New comments are not allowed.